 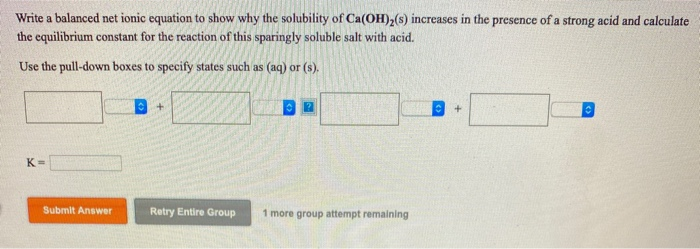
Balancing by mass means making sure that there are equal numbers of each element. Let’s use the reaction between sodium chloride and silver nitrate as an example. There are three main steps for writing the net ionic equation for CH3NH2 + HCl CH3NH3Cl (Methylamine + Hydrochloric acid). Net ionic equations must be balanced by both mass and charge. The first step to writing a net ionic equation is balancing the chemical equation present. It makes it easy to see the active molecules in a reaction, since they are the only ones present in the equation! Writing net ionic equations Why do we use net ionic equations?Īs described above, we use net ionic equations to emphasize the molecules that undergo a change in the reaction. In this equation, the spectator ions are not present. This online chemistry calculator balances equations of chemical reactions. The definition of a net ionic equation is an equation that depicts only the molecules or ions that are actively involved in the reaction or those that undergo a change. Aqueous solution: a solution in which the solvent is water.The ions switch partners because opposites attract. A metathesis reaction is when cations and anions switch partners (double replacement reaction). It allows us to see what caused the solids, liquids, or gases to form. 
Archived chem 102 net ionic reaction of excess CuSO4 with NH3 Answered. A net ionic equation shows exactly what reactants are involved in the reaction. Spectator ion: ions in a reaction that do not undergo any change. Chemical Equation Calculator is a free online tool that displays the structure, balanced equation, equilibrium constant, substance properties with chemical. Normal Balanced Formula: CuSO4 (s) + 2 NH4Cl (aq) CuCl2 (s) + (NH4)2SO4 (aq) Ionic Equation: Net Ionic Equation: Also, could you please explain, how we go from one to another And what are the state symbols Write the state (s, l, g, aq) for each substance.Net ionic equation: a molecular equation that depicts the ions or molecules that experience a change within a reaction.Complete ionic equation: a molecular equation that separates the molecules into their ion forms.For example, the chemical reaction between HCl(aq) and Fe(OH) 3 (s) still proceeds according to the equation: 3HCl(aq) + Fe(OH) 3 (s) 3H 2 O() + FeCl 3 (aq) even though Fe(OH) 3 is not soluble. STEP 1: Write the chemical equation HCl (aq) + NaOH (aq) NaCl (aq) + H 2 O (l) STEP 2: Rewrite by separating the soluble ionic compounds into their dissociated ions H + (aq) + Cl (aq) + Na (aq) + OH (aq) Na + (aq) + Cl (aq) + H 2 O (l) Note that water is not separated because it is not an ionic compound. In addition, you will learn how they are useful for understanding which ions in the solution are active in a reaction and which are spectator ions. Neutralization reactions are one type of chemical reaction that proceeds even if one reactant is not in the aqueous phase. Please let us know how we can improve this web app.In this tutorial, you will learn how to write net ionic equations.  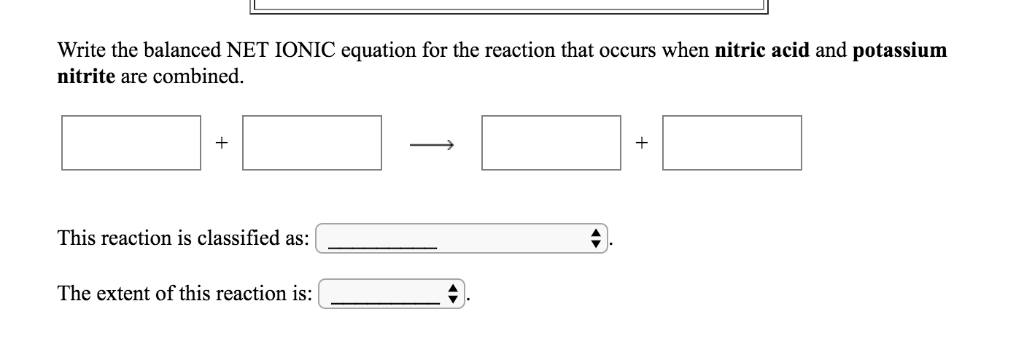
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
